Office of Research & Development |
 |
Office of Research & Development |
 |


Dr. Timothy Hoffman, a radiopharmaceutical chemist, prepares the tracer drug being used to show whether prostate cancer has spread in the Veterans participating in a clinical trial at the Truman VA. (Photo by Mindy Roettgen)
January 12, 2022
By Mike Richman
VA Research Communications
"When used with PET imaging, these radioactive peptides light up on the scan to identify even the smallest amount of disease in its initial growth."
Staging is critical for pinpointing the best way to treat cancer. In staging, doctors determine where the cancer is in the body so they can address it accordingly, whether through surgery, radiation, chemotherapy, or other means.
At the Harry S. Truman Memorial Veterans’ Hospital in Missouri, researchers are testing a new tracer drug for its ability to show whether and where prostate cancer has spread in the body.
Dr. Thomas Dresser, a nuclear medicine physician, and Dr. Timothy Hoffman, a radiopharmaceutical chemist, are carrying out a phase 3 clinical trial on the drug, which contains a pharmaceutical component and a radionuclide component. In the procedure, a prostate tracer drug in a vial is connected to a generator and combines with gallium 68, which emits radioactivity in the form of a positron. That formula, known as Ga68-PSMA-11, is then injected into a patient and binds to the prostate cancer cell because of a protein in the cancer cells called PSMA—prostate specific membrane antigen.
Images from a positron emission tomography (PET) scanner show the location of the cancer.
This type of drug appears to be the best diagnostic tool for staging prostate cancer and determining whether it has metastasized, says Dresser, the chief of nuclear medicine at the Truman VA.
“Once administered intravenously, radiopharmaceutical peptides are attracted to and bind to cancerous cells,” he explains. “When used with PET imaging, these radioactive peptides light up on the scan to identify even the smallest amount of disease in its initial growth. We scan the entire body and can identify prostate cancer with a higher degree of accuracy than a CT scan or a bone scan.
“When a normal cell in the prostate gland becomes cancerous, the membrane of the cell begins to make too many copies of PSMA,” he adds. “This unique cell marker can help us diagnose prostate cancer. However, current imaging does not clearly identify if and where metastasis has occurred. The key to diagnostic precision is the use of this injectable radiopharmaceutical peptide designed for a specific cancer and used in conjunction with PET imaging.”
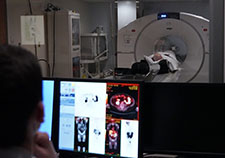
A Veteran undergoes positron emission tomography scanning in the clinical trial. (Photo by Mindy Roettgen)
To date, Dresser and his staff have staged cancer in 25 Veterans at the Truman VA who were diagnosed with prostate cancer through biopsies. The researchers are comparing the findings of PET imaging with those of a CT scan and a bone scan. Many patients in the group of 25 have shown upstaging, which is the effect of finding more disease in this case beyond the prostate and changing the stage, according to Dresser.
“This is important because if someone is treated for limited disease when extensive disease is present, they will not receive the cure that was planned,” Dresser says. “They may return in one year and find the disease has returned when, in fact, it was there from the beginning. It may have spread to the lymph nodes, bones, and other tissue. But the older methods did not detect it. Historically, we’ve not had a good way to determine where it’s spread. PSMA imaging will greatly improve the management of prostate cancer. It’s a game changer.”
Dresser expects to conduct PSMA imaging on about 100 patients over a year.
Prostate cancer is most common in men ages 65 and older. About one out of eight men will be diagnosed with the disease at some point in their life. Cancer of the prostate, a walnut-sized gland that is part of the male reproductive system, is usually found in its early stages, typically due to PSA (prostate specific antigen) screenings. It often grows slowly.
But some cases can become aggressive and spread to other parts of the body. Early detection and treatment are key factors that influence outcomes. However, identifying specific areas where the cancer has spread with traditional means can be challenging.

VA researchers using AI to decide best treatment for rectal cancer

VA center training the next generation of researchers in blood clots and inflammation

AI to Maximize Treatment for Veterans with Head and Neck Cancer

Veterans help find new cancer treatments
The phase 3 trial at the Truman VA is a byproduct of the foresight of Hoffman, who has worked in nuclear medicine research and development for more than four decades at that hospital. Several years ago, he began researching the development of new drugs that can potentially diagnose and treat prostate cancer. He reviewed the cancer screening tools at the Truman VA and wondered whether patients with prostate cancer would be eligible for the radiopharmaceutical Ga68-PSMA-11.
At the same time, Hoffman says, “I realized that the Truman VA was literally five to eight years behind the curve on having access to state-of-the-art diagnostic clinical PET imaging for prostate cancer. I asked, `What’s it going to take for us to do it.’ I thought that as a chemist I can make the [tracer] drug.
“Our local hospital was not making PET drugs and did not have access to those drugs because we’re located in the central part of Missouri in a rural community area,” he adds. “We would have to send our patients at least two to three hours away to get this type of a test done. So I went backward one more step and said, `We’re going to set up a radiopharmacy, and we’re going to start making FDA-approved radiopharmaceuticals for Veterans. Then I’m going to use that expertise and show the FDA that if I can do it for an FDA-approved drug, I should be able to do it for an investigational new drug [IND], and I’ll file the IND for those drugs.’”
A little more than a year ago, the FDA granted approval to the University of California, Los Angeles, and the University of California, San Francisco, to use Ga68 PSMA-11 in conjunction with PET imaging to diagnose prostate cancer. The FDA gave approval only to those two universities, Hoffman explains, because their in-house produced drug proved to be safe and effective. He says the Truman VA is making Ga68 PSMA-11 based on a different formulation, hence the need for the IND. (In December 2021, the FDA granted full approval for use of Ga-68-PSMA-11 in prostate cancer imaging. But Dresser and Hoffman are still evaluating it as a research drug with the same question as the one in their clinical trial: Is PSMA imaging superior to conventional imaging?)
With support from the University of Missouri School of Medicine, where both he and Dresser teach, Hoffman led efforts at the Truman VA to create the radiopharmacy. VA’s Office of Research and Development, specifically its Biomedical Laboratory Research and Development arm, funded the equipment. In August 2021, the Truman VA became the first VA hospital to offer this level of care in-house, marking the beginning of the phase 3 trial.
“We’re not only going to invest in it, but our patients are going to be diagnosed way earlier now because they’re going to know exactly where the cancer is,” Hoffman says. “Up until this point, a man with a slightly elevated PSA … they’d suggest to just watch and see what happens. Now we’re finding in screening that patients with low PSAs have unknown metastatic disease by conventional imaging, meaning a CT scan, bone scan, and a normal workup. This drug is showing that there is metastatic disease, and that it’s time to put these patients on a different treatment protocol.”
Why would a patient whose PSA is only slightly elevated elect to undergo this procedure? PSA levels between 4 and 10 indicate a risk of cancer higher than normal.
“That’s why we’re gathering our data,” Hoffman says, “which are indicating that even at lower PSAs, there are findings that are clinically relevant and that will benefit the long-term survival of these patients.”
Hoffman makes the Ga 68 PSMA-11 in his radiopharmacy and gives it in a syringe to Dresser, who injects it into the patient and performs the PET imaging. The formula has a half-life of only 68 minutes, meaning the drug must be injected into a patient almost immediately after it is prepared.
“The injection goes through the body, and the drug accumulates in the cancer,” Dresser says. “It’s excreted out in the urine, and we see it in salivary glands and the liver. Then we get a picture of the whole body. That shows us where the cancer is. We can make measurements to see how much has accumulated. Then we can measure the uptake value in different tissues and have a scoring system to say this is high probability or low probability.
“In addition to this scan of the body, a concurrent CT scan is used for what we call attenuation correction. We get some anatomic information. When we see it in the pelvis, for instance, we superimpose the CT scan on top of that, so we have two images superimposed. Then we can see exactly where it is. We can see if it’s in a lymph node or a blood vessel. That’s the complete set of images that we look at to make the diagnosis.”
Dresser reports the results to an oncologist or a urologist for treatment recommendation.
“Let’s say we find that the cancer is only in the prostate,” he says. “Now we know the treatment can be focused only in the prostate. Then the urologist will say, `Do you want a radical prostectomy [removal of the prostate]? Here are the advantages and disadvantages to that. Do you want radiation therapy? Here are the advantages and disadvantages to that. Do you want androgen deprivation therapy?’” The latter is a hormone-suppressing therapy used to treat prostate cancer.
“The next patient we evaluate may have some regional lymph nodes. So now the treatment’s going to be different. Removing the prostate is off the books because that’s not going to cure you. You’re going to need additional therapy. The benefit of this test is we can accurately diagnose the stage, so we can give the best treatment for it because we have complete information.”
The most common side effects from Ga68 PSMA-11, nausea, diarrhea, and dizziness, have been seen in less than 1% of the patients studied thus far. The risk of cancer from the radiation is also low, Dresser says, noting that a Ga68-PSMA test emits a level or radiation just above that of a standard CT of the chest or abdomen. “I expect that future evaluations of patients with diagnoses of prostate cancer will include only the PSMA test and not the conventional imaging of a bone scan or a CT scan,” he says. “Therefore, the risk of radiation will be same as it is now.”
Hoffman is confident that Ga68 PSMA-11 has the potential to trace other types of cancer. He’s in discussions with Dresser to use the drug for patients with hepatocellular cancer, which is linked to hepatitis C, a liver infection that, like prostate cancer, is a major health issue among VA patients. In recent years, studies in the United States and Europe have supported the potential of Ga68 PSMA-11 imaging in hepatocellular cancer. Plus, Hoffman’s radiopharmacy is producing an FDA-approved diagnostic agent called Netspot in conjunction with Ga68 PSMA-11 to identify and diagnose neuroendocrine tumors, which can occur anywhere in the body and can also signal pancreatic cancer.
"Our capability to prepare Ga-68 PET radiopharmaceuticals now opens the door for all future Ga-68 drugs to be prepared on-site and to be readily available," Hoffman says. "This is a big development that creates much promise for cancer imaging down the road when it comes to primary screening in VA hospitals."
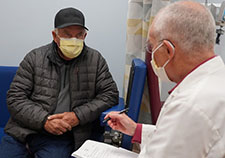
Nuclear medicine physician Dr. Thomas Dresser (right) consults with Air Force Veteran Kenneth Stufflebean. PET images showed that prostate cancer has spread to two lymph nodes in Stufflebean's pelvis. (Photo by Mindy Roettgen)
Air Force Veteran Kenneth Stufflebean is participating in the phase 3 clinical trial at the Truman VA that is aimed at improving the ability to stage prostate cancer.
After a blood test showed an elevated PSA score and a biopsy of his prostate showed extensive cancer, Stufflebean underwent conventional testing with a bone scan and a CT scan to identify the location of the cancer. Both tests showed it was concentrated only in his prostate.
That meant the stage of his cancer would normally be llA, or one-half or less of one side of the prostate. The recommended treatment could be a radical prostatectomy, which is removal of the prostate, or radiation only to that gland.
But Stufflebean and his doctors took it one step further. After an injection of the radioactive prostate cancer tracer drug being tested in the clinical trial—Ga68-PSMA-11—he underwent PET imaging (see main story). The images showed that the cancer had spread to two lymph nodes in the pelvis, the area below the abdomen that is located between the hip bones and contains the bladder and rectum.
Based on the PET images, the stage of his cancer appeared to be IVA, meaning it had spread to lymph nodes near the internal iliac artery, which sends blood to the pelvis.
“Although we do not have tissue confirmation, the likelihood for disease is very high,” says Dr. Thomas Dresser, a nuclear medicine physician who is co-leading the trial. “Based on this information, his treatment is going to be different—probably radiation of the pelvic area.”
Stufflebean, 75, served in the Air Force from 1966 to 1970, spending time stateside and in Thailand. He lives about 100 miles from the Truman VA in Marceline, Missouri, but originally thought about traveling to the University of California, Los Angeles—one of two schools approved by the FDA in 2020 to carry out the PET imaging technique using Ga68-PSMA-11.
After consulting with Dresser, who explained to him that the Truman VA had acquired FDA approval to experiment with the drug on an investigational basis, he decided to undergo the procedure right there.
Stufflebean is glad that additional testing found the cancer in his lymph nodes so the treatment plan could be adjusted accordingly.
“I’m very pleased that they found it in the lymph nodes,” he says. “I’ve been going to the VA since 2008. They’ve run a lot of lab tests on me. If it hadn’t been for just giving a [PSA] blood sample, I wouldn’t have known there was anything wrong with me. You don’t go to the doctor if you feel good. I’ve got no pain, but it’s hard for me to control my bladder. The blood test is what showed it right off the bat.”
He expects to begin radiation treatment around the beginning of February.
“I’m anxious to get this started, but I’m just going to do what they tell me to do,” he says.
--- Mike Richman
VA Research Currents archives || Sign up for VA Research updates