Office of Research & Development |
 |
Spinal cord injuries (SCIs) impair the brain's ability to send messages to the rest of the body. These injuries can result in paralysis, loss of feeling, chronic pain, and other serious medical problems below the site of the injury.
SCIs are estimated to affect between 249,000 and 363,000 Americans, with about 17,730 new injuries occurring each year. About 80% of people with new injuries are males, and roughly 42,000 people with SCIs are Veterans. Nearly half of all SCIs occur in people between the ages of 16 and 30, so many patients live with the effects of these injuries for decades.
VA provides care to more than 27,000 Veterans with SCIs and related disorders (SCI/D) each year, making the department the largest health care system in the world providing lifelong spinal cord care. VA integrates vocational, psychological, and social services, and addresses the changing needs of Veterans with SCI/Ds throughout their lives.
SCI services are delivered through a "hub and spoke" system of care, extending from 25 regional SCI centers that offer primary and specialty care by multidisciplinary teams to more than 130 SCI primary care teams or support clinics at local VA medical centers. Locations and telephone numbers for each of VA's SCI center can be found here.
VA is also one of 23 members of the Consortium for Spinal Cord Medicine, an organization founded by Paralyzed Veterans of America (PVA) to make care for persons with SCI/D more evidence-based. The consortium makes recommendations to health care providers based on current research findings that experts have graded for their scientific strength and validity.
The VA Advanced Platform Technology (APT) Center focuses on the practical medical needs of Veterans disabled by problems related to the sensory or motor systems, cognitive deficits, or limb loss. Their work emphasizes prosthetics and orthotics, wireless health monitoring and maintenance, and interfaces to connect the nervous system with internal or external devices. Their goal is to harness emerging technologies to enable people with SCI or other neurological deficits to exert greater control over their bodies.
VA’s Center for Functional Electrical Stimulation (FES) in Cleveland focuses on the application of electrical currents to either generate or suppress activity in the nervous system through functional electrical stimulation. FES can control the movement of otherwise paralyzed limbs for standing and hand grasp and activate body functions such as bladder control or respiration. Established in 1991, the FES Center is a consortium with three institutional partners: the Cleveland VA Medical Center, Case Western Reserve University, and MetroHealth Medical Center. The Center was founded to introduce FES into clinical practice.
The Human Research Engineering Laboratories (HERL) is operated jointly by the VA Pittsburgh Healthcare System and the University of Pittsburgh. HERL's mission is to continually improve the mobility and function of people with disabilities through advancing engineering and clinical research in medical rehabilitation. HERL features state-of-the-art research instruments and machines for prototype fabrication.
VA research on SCI focuses on returning motor and sensory function to Veterans with these injuries. Researchers look at areas including neural engineering, which designs solutions to problems associated with neurological limitations and dysfunction; wheelchair and other adaptive technology; treatment of the medical complications of SCI; and new rehabilitation methods and outcomes.
VA researchers are also working in the field of regenerative medicine, learning how to restore tissue and organ function, including spinal cord function, lost as a result of aging, injury, or disease. They are using a variety of tools to change the ways wounds heal in humans to enable damaged organs and tissues to repair and regenerate.
If you are interested in learning about joining a VA-sponsored clinical trial, visit our research study information page.
New consortium to restore lost function—Researchers are pursuing new methods to repair damage to the spinal cord and restore function after SCI. In 2010, VA and several academic partners established a consortium, now named the Gordon Mansfield SCI Consortium, to pursue high-risk, high-return ideas that might otherwise be unlikely candidates for funding.
The consortium hopes to advance the field of regenerative rehabilitation. Scientists in the consortium have already generated promising early findings highlighting cell transplantation as a potential way to repair the nervous system. They have demonstrated in animal models that cell transplants can survive and integrate with a chronically injured spinal cord and form connections with host tissue.
Cellular therapies to restore spinal cord function include stem cell transplantation, tissue engineering, the use of biomaterials, and genetically manipulated cells to repair damaged or diseased tissues. Researchers are using tissue engineering to manufacture temporary scaffolds (a framework or structural element used to hold cells together) that contribute to the formation of new functional tissue.
Transplanting neural cells in the spinal cord—In 2019, researchers from the VA San Diego Healthcare System and the University of California San Diego found that neural stem cells (immature stem cells in the brain that can become more specialized) act like the cells of normal spinal cords when they are grafted into the locations of spinal cord injuries in rats.
As the stem cells develop, they spontaneously segregate themselves into cell clusters, some supporting movement and others supporting feeling. In addition, they reproduce into a variety of cells found in normal spinal cords and find appropriate places to locate within the graft. The study demonstrates that it may be possible to use stem cells to form a “bridge” around an injury to restore both movement and feeling in people with SCIs.
3D printed spinal cord—Another 2019 study from the VA San Diego Healthcare System and the University of California San Diego describes how researchers used rapid 3D-printing technologies to create a spinal cord facsimile. The team loaded the scaffold with neural stem cells and successfully implanted it into sites of severe SCI in rats.
The 3D-printed scaffolds reproduce the slender, bundled arrays of axons (a portion of the nerve cell that conducts electrical impulses) in the spinal cord, helping stem cells to replicate the anatomy of the spinal cord before it was injured. The scaffolding keeps axons in order, guiding them to grow in the right direction to complete a spinal cord connection.
The use of 3D printing allows clinicians to quickly print out an implant that will match the injured site of a host spinal cord, regardless of its size and shape. Rats treated in this way regained significant function in their hind legs after a few months—and new spinal cord tissue grew across the injury and connected the severed ends of the host spinal cord.
Helping nerve fibers regenerate—Boosting energy levels within damaged nerve fibers (axons) may help them regenerate and aid in the recovery of function, according to a 2020 study by a team of researchers from the Richard L. Roudebush VA Medical Center in Indianapolis and Indiana University. Cells produce energy through the action of tiny power plants called mitochondria, which convert nutrients and oxygen into fuel.
In mouse models, injured axons do not regenerate because of energy deficits and mitochondrial dysfunction. The team found that increasing function and energy metabolism within the mitochondria is key to enabling axons to regenerate and improve recovery of function after SCI.
In three mouse model experiments, the team stimulated mitochondria by deleting a protein anchor called syntaphilin and increased energy metabolism through creatine treatment. This strategy, the team believes, can be translated into future SCI treatments.
Bioengineered spinal discs—In 2018, researchers at the Corporal Michael J. Crescenz VA Medical Center in Philadelphia and the University of Pennsylvania successfully implanted bioengineered spinal discs into goats.
The treatment combined an animal's stem cells with biomaterial to generate a composite structure that was implanted into its spine to act as a replacement spinal disc. Goats were chosen because their cervical spinal disc dimensions are similar to humans and they have a semi-upright stature. Researchers found the implanted discs' mechanical properties either matched or exceeded those of native goat cervical discs, eight weeks after the procedure.
The success of the study offers “compelling” evidence that tissue-engineered analogs might soon be used to replace a deteriorated spinal disc in humans. The team is optimistic they can achieve the same level of success in human trials.
Denosumab is a monoclonal antibody drug that is used to treat osteoporosis, bone loss, and problems related to bone cancer. Researchers at the James J. Peters VA Medical Center in New York found it can be used to preserve bone density in patients with SCIs, according to a 2020 study.
Patients with SCIs are at high risk of osteoporosis and bone fractures because of sluggish blood flow, hormonal alterations, and other factors. Without strong bones to support the body, patients can have difficulties using exoskeletal devices or spinal cord stimulation to help them walk, which could result in fractures.
The research team found SCI patients who received denosumab maintained higher bone mineral density at the knee 18 months after treatment than those who received a placebo.
Brief exposures to hypoxic air (air that contains reduced oxygen levels), interspersed with periods of breathing room air, can improve motor function in people with SCI, found a 2020 article by researchers at the Edward Hines Jr. VA Hospital in Illinois and the University of Miami.
The team found that acute intermittent hypoxia (AIH) paired with transcranial magnetic stimulation increased electrical activity in muscles within the hand, leading to improved motor output in patients with intact spinal cords. The researchers concluded the treatment’s ability to induce temporary changes in neuron excitability can help patients with SCI recover lost motor function. The group is continuing its efforts to understand how AIH supports human motor systems.
Implanted neuroprosthetic devices—In 2018, researchers at VA's Advanced Platform Technology (APT) Center and Case Western Reserve University published the results of a 10-year clinical trial to test a surgically implanted electrical stimulation system in people with SCI. During the surgery, electrodes were implanted in muscles located in the subjects' trunk and legs. Electrical leads were then connected to a neurostimulator. The team found that implanted lower-limb neuroprostheses can provide lasting benefits for recipients.
By stimulating muscles, neuroprostheses promote improved standing, balance, and the capability for exercise. Patients are given functional training on how to use the stimulation system and are prescribed a course of exercise. Lab tests focus on strength, balance, and function with and without the system.
In 2019, researchers at the Cleveland FES Center announced they had developed a method to activate abdominal and lower rib cage muscles to produce an effective cough, using minimally invasive techniques. Electrodes were inserted under the skin and then advanced to the dorsal spinal cord. Research participants used a stimulator to produce several different cough efforts ranging from light to strong. It is the first method in the world that can restore cough to SCI patients.
Preventing falls from wheelchairs—To improve wheelchair safety, researchers with the APT Center developed an algorithm to detect sudden stops during wheelchair use. Sudden stops can often cause falls from the wheelchair, especially for users who are paralyzed. The team paired the algorithm with a neurostimulation device that increases trunk stability, reducing the likelihood that the user will be dislodged from the wheelchair during a sudden stop.
Researchers tested the system on three volunteers with SCI, who reported feeling safer. The study, published in 2020, suggests that neurostimulation systems can improve wheelchair safety for paralyzed users.
Electronic stimulation safety—APT Center researchers showed, in a 2020 study, that nerve health is maintained after implanting an electrode stimulation device in a patient with SCI. A patient with leg paralysis was given an implanted electrode device to stimulate his leg muscles and help him to stand.
The researchers examined physiologic changes to the patient's nerves pre-surgery, post-surgery, and at one-year follow up. They found minimal nerve irritation during surgery that resolved after the procedure. Diagnostic testing found preserved nerve health with strengthened responses following stimulated exercise, demonstrating that implanted electrodes to restore function after SCI can be both safe and effective.
Transcranial direct current stimulation—In a pilot study published in 2018, a team of researchers from the APT Center and the Cleveland Clinic found that transcranial direct current stimulation (tDCS) holds promise to restore function in SCI patients.
The team investigated the combined effects of tDCS and rehabilitation on Veterans with long-term, incomplete SCI that affected upper-limb function. In tDCS, electrical currents are applied directly to the brain. The technique is similar to transcranial magnetic stimulation (TMS), a non-invasive procedure used to treat major depressive disorder and other conditions.
Study participants who received tDCS and rehabilitation for two weeks had increased muscle strength in their hand, wrist, and proximal arm muscles that lasted for three months, compared to a group that received a sham treatment.
Low-dose testosterone replacement therapy paired with resistance training can lead to improved muscle mass and health in SCI patients, according to a 2019 study by researchers at the Hunter Holmes McGuire VA Medical Center in Richmond, Virginia.
Patients who took part in the study underwent twice-weekly resistance training paired with electrical stimulation for 16 weeks, to activate paralyzed muscles in their legs. They were also given testosterone replacement therapy through a patch. Patients who received both testosterone and resistance training had improved body lean mass, muscle mass, and knee extensor strength. They also had, overall, less body fat, an increased metabolic rate, better ability to process glucose, and a stronger inflammatory response.
The researchers concluded that low-dose testosterone replacement therapy can ease the negative muscle and metabolic effects of SCI.
Smart bandage technology—Researchers at the APT Center have developed a "smart bandage" that makes use of electrical stimulation to treat chronic wounds that will not heal on their own. Chronic pressure injuries frequently occur in people with SCI. The layered smart bandage includes an absorbent material to protect the wound, a mechanism to deliver electrical stimulation, temperature sensors, and a smart chip.
Electrical stimulation provides energy to promote healing in a chronic wound. The temperature sensor helps clinicians monitor wound healing without having to remove the bandage. The bandage can last for up to seven days. If needed, a new bandage can be affixed to the existing electronics for another seven days, or for as long as the patient requires.
Pressure injuries can be expensive to manage, both for the individual and the health care system. The new bandage could change that dynamic, making it possible for clinicians to remotely monitor wound healing from the patient's home, reducing clinic visits. The research team is working with VA's Technology Transfer Program and outside entrepreneurs to bring the invention to market.
New pressure injury measurement tool—An innovative computer-based system to more accurately measure pressure injuries is being tested by researchers at the James A. Haley Veterans Hospital in Tampa, Florida.
The system captures 3D-images with a camera mounted to a tablet. An algorithm uses the color, geometry, and formation of the wound to assess its length, width, and depth. The research team believes the new tool is more reliable than the current pressure ulcer monitoring tool.
Accurate pressure injury measurement is vital to allow clinicians to accurately assess the severity of the wound and the degree of tissue damage, and to determine whether current treatment strategies are effective. Intervening early to prevent injuries from worsening is critical for improving patient outcomes and reducing costs. Following the completion of a preliminary trial in 2018, the team hopes to assess the effectiveness of the new system compared with manual pressure injury measurements.
BrainGate—VA has played a major role in supporting the establishment of BrainGate, a collaborative research enterprise that seeks to develop new neurotechnologies. Researchers at the Providence VA Medical Center in Rhode Island, Brown University, and other institutions spearheaded the development of brain-computer interfaces that rely on microelectrodes implanted in the brain. The electrodes send signals to an external decoder that translates them into commands for robotic devices, helping users to restore lost function and control over their environment.
Among other projects, the BrainGate team is now studying whether the system can be effective as a means of intuitive control of prosthetic limbs or as a way to help patients move their own paralyzed limbs.
A 2011 study found the BrainGate system continued to control a computer cursor accurately through brain activity for more than 1,000 days after it was initially implanted. In 2012, in a widely publicized study, the team showed that people with tetraplegia (paralysis that affects the upper and lower body) could use the system successfully to control a robotic arm to reach and grasp for items.
ReHAB—The FES Center and the consortium are currently working on a system called ReHAB (Reconnecting the Hand and Arm to the Brain). The system uses an array of sensors surgically implanted into different areas of the brain along with electrodes placed directly on nerves in the hand, arm, and shoulder. The device detects paralyzed users' thoughts and then stimulates nerves in the upper limbs to initiate movement. An early feasibility study of this system is scheduled to be completed in 2024.
Restoring lost nervous system function—The VA Center for Neurorestoration and Neurotechnology (CfNN), located in Providence, Rhode Island, aims to advance and translate neurotechnology to restore lost function. Through multiple areas of focus, CfNN seeks to develop, test, and implement new therapies and technologies that restore function for Veterans with disorders affecting the nervous system.
In 2018, a team that included researchers from the CfNN showed that “local field potentials”— the total electrical current flowing to a small section of nervous tissue in the brain—may be a more robust, stable signal for brain-computer systems than neuronal action potentials, which represent the activity of individual brain cells.
The VA Rehabilitation Research and Development Center for the Restoration of Nervous System Function (CRNF) in West Haven, Connecticut, is dedicated to making clinical, molecular and cell-based discoveries to alleviate pain and restore nervous system function in Veterans whose nerves have been damaged by injury or illness such as SCI, multiple sclerosis, and diabetes.
Studies at the CRNF center focus on the role of sodium channels in mediating chronic neuropathic pain in various diseases. Sodium channels are proteins that conduct sodium ions through a cell's plasma membrane. By transmitting impulses quickly throughout cells and cell networks, sodium channels enable coordination of higher processes ranging from movement to cognition.
In 2019, a group of researchers from the CRNF and other institutions identified a mutation in α-subunits of voltage-gated sodium channels that is responsible for chronic neuropathic pain in diabetes patients.
VA's Center on the Medical Consequences of Spinal Cord Injury is located at the James J. Peters VA Medical Center in New York. The center's mission is to improve Veterans' quality of life and increase their lifespan by preventing consequences from an SCI. These secondary injuries can include bone and muscle loss, as well as metabolic and cardiovascular changes.
Researchers at the center continue to study an Israeli technology that allows paraplegics to stand, walk, and climb stairs, called ReWalk. ReWalk is a wearable robotic exoskeleton that provides powered hip and knee motion to enable individuals with SCI to stand upright, walk, and turn. On their first day using the device, most people can stand and take a few steps, although it takes practice and training to use it properly. The FDA approved the ReWalk exoskeleton for sale in the United States in 2014, and VA now purchases the apparatus for eligible Veterans with SCIs.
VA is currently conducting a four-year clinical trial to evaluate the impact of exoskeleton use in home or daily life settings. The 160-person trial is scheduled to be completed in 2021.
The Human Research Engineering Laboratories (HERL), operated jointly by the VA Pittsburgh Healthcare System and the University of Pittsburgh, aims to improve mobility and function in people with disabilities through advanced engineering.
HERL is responsible for multiple innovations in wheelchair design and other assistive technologies that have changed the way hundreds of thousands of Veterans interact with their environment and approach daily living.
The team has 25 patents in wheelchair design and technology to its credit. Examples of their cutting-edge innovations include a waterproof wheelchair that runs on compressed air; a robotic wheelchair called MEBot; a powered wheelchair fitted with robotic arms to help users with limited hand function; and a SmartWheel device that evaluates manual wheelchair use by measuring push forces, smoothness, and speed.
In 2020, researchers from HERL and the University of Pittsburgh published findings on new designs for an adjustable wheelchair that allows users to participate in para-table tennis events. The team found that specific support, movements, and posture needed to competitively participate in table tennis are different than general day-to-day mobility requirements.
Acute intermittent hypoxia boosts spinal plasticity in humans with tetraplegia. Christiansen L, Chen B, Lei Y, Urbin MA, Richardson MSA, Oudega M, Sandhu M, Rymer WZ, Trumbower RD, Mitchell GS, Perez MA. Brief exposures to hypoxic air interspersed with periods of breathing ambient room air improve motor function in people with SCIs. Exp Neurol. 2021 Jan;335:113483.
Dried plum mitigates spinal cord injury-induced bone loss in mice. Liu X, Liu M, Truner R, Iwaniec U, Kim H, Halloran B. Dietary supplementation with dried plum can prevent bone loss in an acute model of SCI, and restore a fraction, but not all, of the bone that has already been lost in mice following SCI. JOR Spine. 2020 Dec;4(3):e1113.
Neural stem cell grafts form extensive synaptic networks that integrate with host circuits after spinal cord injury. Ceto S, Sekiguchi KJ, Takashima Y, Nimerjahn A, Tuszynski MH. Highly specialized grafts of neural stem cells integrate with host networks and behave much like neurons in a healthy, undamaged spinal cord. Cell Stem Cell. 2020 Sep 3;27(3):430-440.e5.
Sudden stop detection and automatic seating support with neural stimulation during manual wheelchair propulsion. Foglyano KM, Lombardo LM, Schnellenberger JR, Triolo RJ. A neurostimulation system developed by VA researchers can improve wheelchair safety for paralyzed users. J Spinal Cord Med. 2020 Aug 14;1-10.
Chronic nerve health following implantation of femoral nerve cuff electrodes. Freeberg MJ, Pinault GCJ, Tyler DJ, Triolo RJ, Ansari R. Nerve health is maintained after implanting an electronic stimulation device in a patient with SCI. J Neuroeng Rehabil. 2020 Jul 14;17(1):95.
Start-up propulsion biomechanics changes with fatiguing activity in persons with spinal cord injury. Bossuyt FM, Hogaboom NS, Worobey LA, Koontz AM, Arnet U, Boninger ML. Investigating changes in wheelchair start-up propulsion biomechanics with fatigue could provide insight into shoulder injury risk. J Spinal Cord Med. 2020 Jul;43(4):476-484.
Administration of denosumab preserves bone mineral density at the knee in persons with subacute spinal cord injury: findings from a randomized clinical trial. Cirnigliaro CM, La Fountaine MF, Parrott JS, Kirshblum SC, McKenna C, Sauer SJ, Shapses SA, Hao L, McClure IA, Hobson JC, Spungen AM, Bauman WA. Denosumab can be used to preserve bone density in spinal cord injured patients. JBMR Plus. 2020 Jun 25;4(8):e10375.
Comparison of carbon fibre and aluminium materials in the constriction of ultralight wheelchairs. Gebrosky B, Grindle G, Cooper RM, Cooper RA. Carbon fiber wheelchair construction is a viable alternative to aluminum, titanium, or steel construction, and decreasing costs will continue to improve the benefits of carbon fiber over these models. Disabil Rehabil Assist Technol. 2020 May;15(4):432-441.
Restoring cellular energetics promotes axonal regeneration and functional recovery after spinal cord injury. Han Q, Xie Y, Ordaz JD, Huh AJ, Huang N, Wu W, Liu N, Chamberlain KA, Sheng ZH, Xu XM. Boosting energy levels within damaged nerve fibers or axons may represent a novel therapeutic direction for axonal regeneration and functional recovery. Cell Metab. 2020 Mar 3;31(3):623-641.
Wrong site spine surgery in the Veterans Administration. Watts BV, Rachlin JR, Gunnar W, Mills PD, Neily J, Soncrant C, Paull DE. Wrong level surgery of the spine is a significant safety issue facing the field that continues to occur despite surgical teams following guidelines. Clin Spine Surg. 2019 Dec;32(10):454-457.
Rehabilitation Engineering: A perspective on the past 40-years and thoughts for the future. Cooper RA, Cooper RM. Over the past four decades, there have been breakthroughs in communication, mobility, and tools to manipulate objects. Probably the most important transformation has been the growing inclusion of people with disabilities into the prioritization, conceptualization, and design of new assistive devices. Med Eng Phys. 2019 Oct;72:3-12.
Low-dose testosterone and evoked resistance exercise after spinal cord injury on cardio-metabolic risk factors: An open-label randomized clinical trial. Gorgey AS, Khalil RE, Gill R, Gater D, Lavis TD, Cardozo CP, Adler RA. Low-dose testosterone replacement therapy paired with resistance training can lead to improved muscle mass and health in spinal-cord-injured patients. J Neurotrauma. 2019 Sep 15;36(18):2631-2645.
The Modular Adaptive Electrotherapy Delivery System (MAEDS): an electroceutical approach for effective treatment of wound infection and promotion of healing. Bogie KM. The MAEDS electrotherapy “smart bandage” can significantly inhibit infection and enhance healing rate in acute infected wounds. Mil Med. 2019 Mar 1;184(Suppl 1):92-96.
Regenerating corticospinal axons innervate phenotypically appropriate neurons within neural stem cell grafts. Kumamaru H, Lu P, Rosenzweig ES, Kadoya K, Tuszynski MH. Neural stem cells act like normal spinal cord cells when transplanted into mice. Cell Rep. 2019 Feb 26;26(9):2329-2339.e4.
Biomimetic 3D-printed scaffolds for spinal cord injury repair. Koffler J, Zhu W, Qu X, Platoshyn O, Dulin JN, Brock J, Graham L, Lu P, Sakamoto J, Marsala M, Chen S, Tuszynski MH. Three-dimensional biomimetic scaffolds offer a means of enhancing central nervous system regeneration through precision medicine. Nat Med. 2019 Feb;25(2):263-269.
What is the functional relevance of reorganization in primary motor cortex after spinal cord injury? Urbin MA, Royston DA, Weber DJ, Boninger ML, Collinger JL. A deeper understanding of the pathophysiological mechanisms of motor dysfunction after spinal cord injury is necessary to develop and optimize repair strategies. Neurobiol Dis. 2019 Jan;121:286-295.
Transcranial direct current stimulation (TDCS) paired with massed practice training to promote adaptive plasticity and motor recovery in chronic incomplete tetraplegia: A pilot study. Potter-Baker KA, Janini DP, Lin YL, Sankarasubramanian V, Cunningham DA, Varnerin NM, Chabra P, Kilgore KL, Richmond MA, Frost FS, Plow EB. Transcranial direct current stimulation may be able to restore function in patients with SCI. J Spinal Cord Med. 2018 Sep;41(5):503-517.
Restorative effects of human neural stem cell grafts on the primate spinal cord. Rosenzweig ES, Brock JH, Lu P, Kumamaru H, Salegio EA, Kadoya K, Weber JL, Liang JJ, Moseanko R, Hawbecker S, Huie JR Havton LA, Nout-Lomas YS, Ferguson AR, Beattie MS, Bresnahan JC, Tuszynski MH. Human neural progenitor cells can be successfully grafted into rhesus monkeys with spinal cord injuries. Nat Med. 2018 May;24(4):484-490.
Restoration of reaching and grasping movements through brain-controlled muscle stimulation in a person with tetraplegia: a proof-of-concept demonstration. Ajiboye AB, Willett FR, Young DR, Memberg WD, Murphy BA, Miller JP, Walter BL, Sweet JA, Hoyen HA, Keith MW, Peckham PH, Simeral JD, Donoghue JP, Hochberg LR, Kirsch RF. First report of a combined implanted functional electrical stimulation and intracortical brain-computer interface for restoring both reaching and grasping movements to people with chronic tetraplegia due to SCI. Lancet. 2017 May 6;389(10081):1821-1830.

Download PDF
 A new way to prevent pressure injuries
A new way to prevent pressure injuries
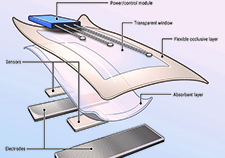 VA researcher develops 'smart bandage' technology for chronic wounds
VA researcher develops 'smart bandage' technology for chronic wounds
 Adaptive rock climbing has physical, psychological benefits for people with disabilities
Adaptive rock climbing has physical, psychological benefits for people with disabilities
 3D images, algorithms are key in potential new tool for measuring bedsores in spinal cord injury
3D images, algorithms are key in potential new tool for measuring bedsores in spinal cord injury